In the regulated life sciences environment, the management of controlled documents such as SOPs (Standard Operating Procedures), procedural instructions or work instructions is of great importance. Change management processes ensure that these documents are properly revised, approved, trained, distributed and, where necessary, suspended. In addition to well-known use cases within change management, there are special cases that are handled differently from company to company.
One of these applications is the rare case of so called controlled printing.
Case Study
A company’s own document management system (DMS) generally distributes controlled documents electronically. Work instructions are electronically implemented in the DMS and then made available to the relevant specialist departments and companies by means of an electronic publishing procedure.
It is not always sufficient to make controlled documents such as work and manufacturing instructions available exclusively electronically. In production plants, critical documents must also be accessible to employees who have no access to electronic media. In particular, batch reports are often filled out by hand in production and then scanned in. In such cases it is still necessary to print documents. Due to the sometimes high criticality of these documents, it must be ensured that no outdated document versions are used, that the printed documents are destroyed after they have been invalidated, and that the entire printing process is comprehensibly documented. This is called controlled printing.
Solution from the user’s point of view (excerpt)
The following chapter describes a simple example of a controlled printing process based on OpenText Documentum for Life Sciences.
A print coordinator requests a controlled printout of a manufacturing instruction. Subsequently, a production plant will produce a single batch of active ingredient using the manufacturing instructions.
The DMS first ensures that only valid versions of the manufacturing instructions are allowed to be printed. The DMS then offers the print coordinator a list of printers in which special paper is inserted (color, watermark, etc.).
Since this is a printout of a manufacturing instruction for production, the print coordinator must still specify the batch number. It is obtained from a third party system, such as a Manufacturing Execution System (MES):
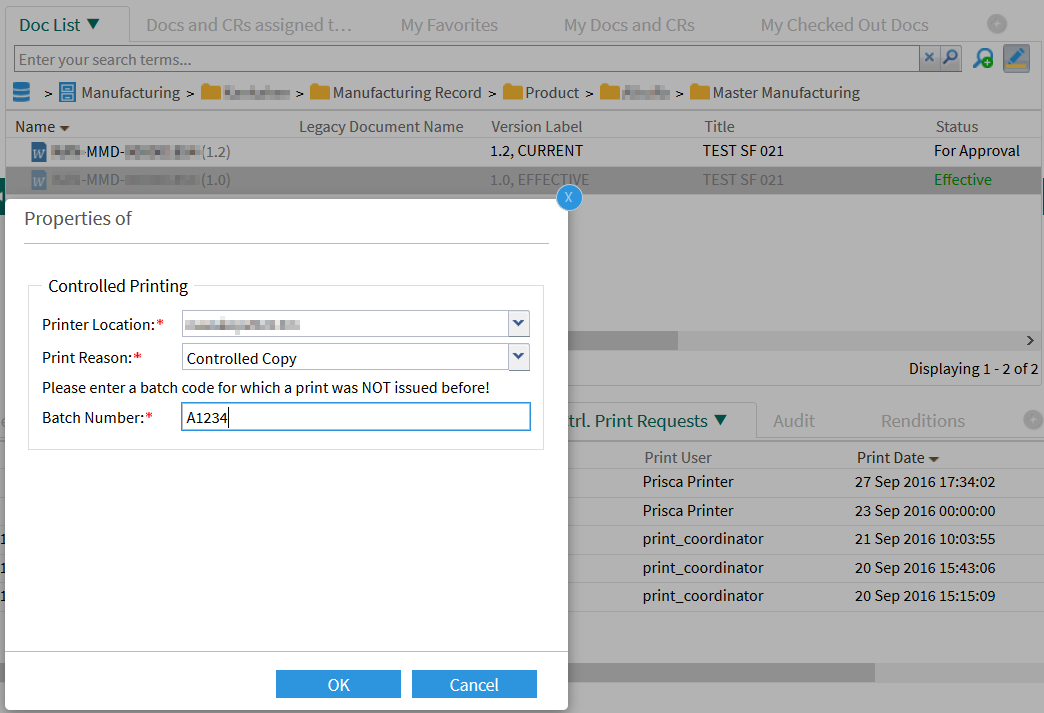
The DMS displays the started printing process:
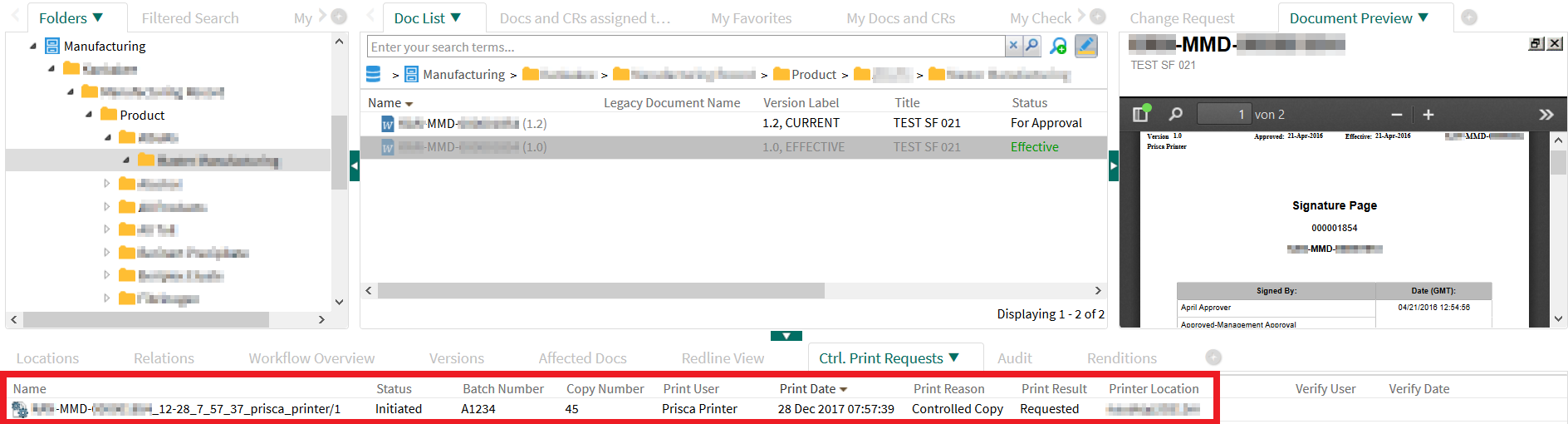
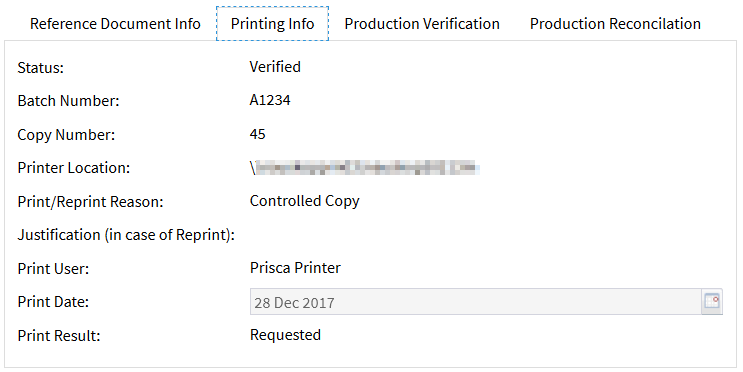
(The following can be identified: name of the print coordinator, total number of paper prints that have been requested so far, date and time, reason, printer hardware.)
After the print coordinator checked the paper printout for completeness and correctness, he confirms this electronically in the DMS:
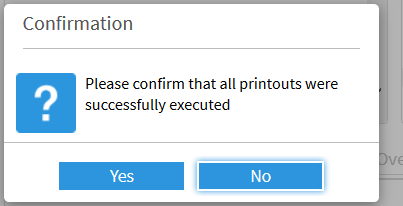
The DMS changes the status of the printing process accordingly:

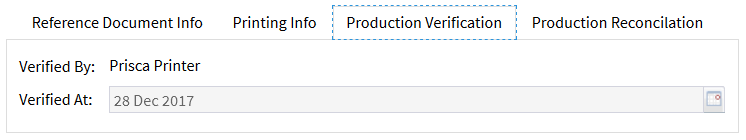
The document coordinators can view details of the printing process at any time:
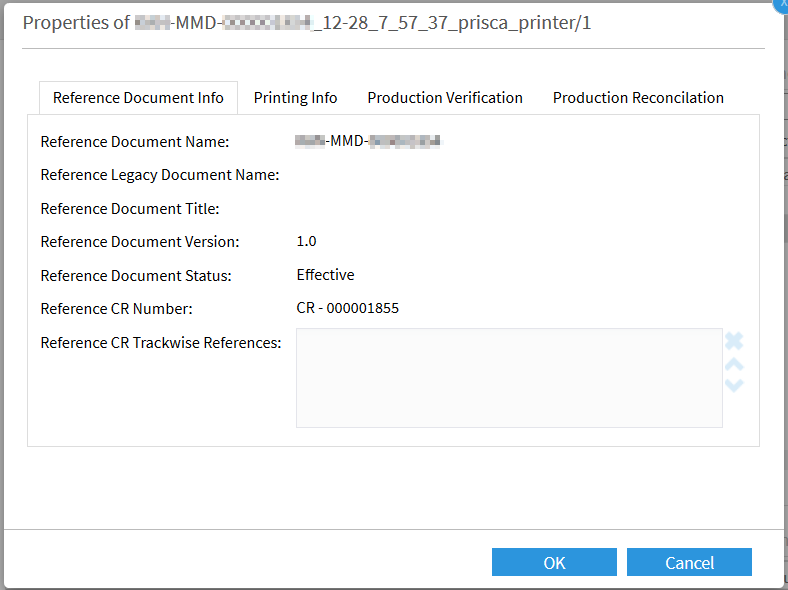
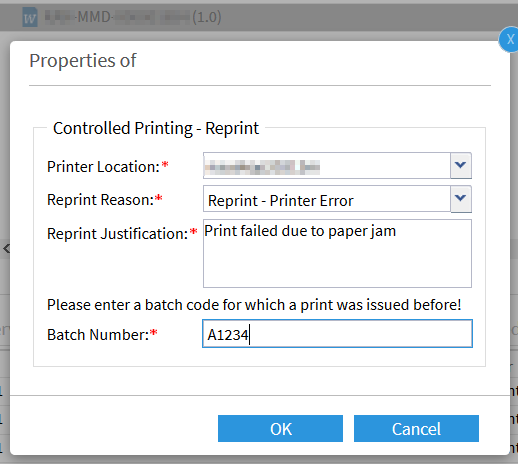
If the printing process fails, the print coordinator can repeat the printing process by entering the same batch number. A justification is necessary and is recorded in an audit trail at all times:
After the printed document has been distributed to the production plant, it may become apparent that an incorrect document has been inadvertently used. In this case, the print coordinator can initiate a recall process:
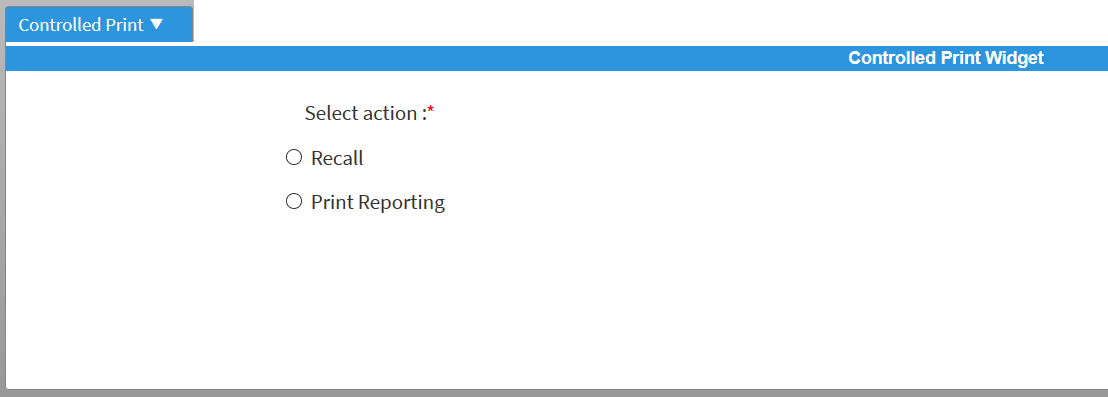
The print coordinator specifies who was the recipient of the document and the reason for retrieving the paper printout:
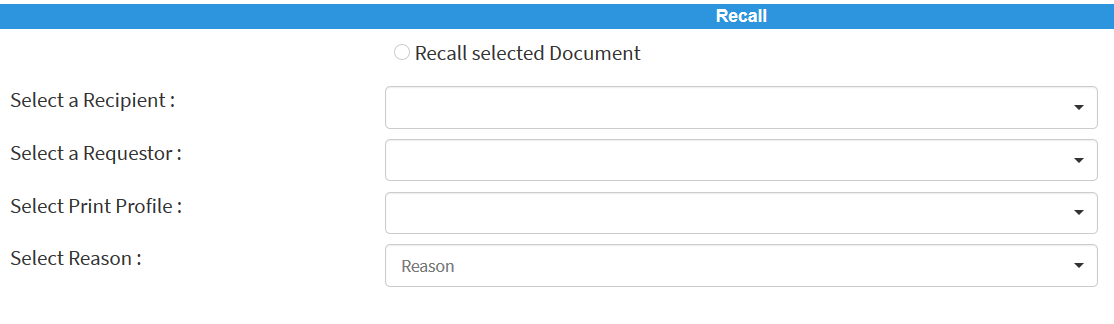
The DMS sends a notification to either a document coordinator or a person responsible in the production plant. Depending on the customer, the printouts are either sent back to the print coordinator or destroyed. He must document both in the DMS and sign electronically.
Do you have any questions or wish further information?
Simply fill in our contact form or visit our OpenText Documentum for Life Sciences landing page.
Here you can find part 1 and 2 of my blogpost series »Special use cases based on OpenText Documentum for Life Sciences«:
{{cta(’59acaef5-2729-46a2-9be9-52f39c573849′)}}





0 Comments